Cannabigerol (CBG) is a phytocannabinoid—i.e. one that occurs naturally in plants, as opposed to produced within the body (known as endocannabinoids), or synthesised cannabinoids. CBG is found in higher concentrations in hemp strains, and is usually present in relatively low levels in drug cannabis varieties.
Cannabigerol (CBG) is one of many phytocannabinoids found in the cannabis plant. Although CBG is not as prevalent in the cannabis plant as THC and CBD, it is a vital chemical precursor to these much-loved cannabinoids.
CBG is more prevalent in the hemp plant, and some geneticists are experimenting with cultivating strains of hemp high in CBG. It has medicinal, non-psychoactive properties of its own, and is being researched for its medicinal characteristics.

CBG & the endocannabinoid system
As a non-psychoactive compound, it is believed that CBG acts as a mild CB1-receptor antagonist, a receptor ligand that binds to the receptor and prevents agonists such as THC from exerting an effect. This is a quality that is observed in the more famous cannabinoid, CBD.
As well as this, CBG has been shown to exert an effect on the 5HT1A-receptor, which assists in the regulation of serotonin release. Cannabigerol moderately blocks the 5HT1A-receptor, therefore affecting serotonin signalling in the central nervous system. CBG is also said to affect the adrenoceptor, therefore controlling expression of adrenaline and noradrenaline in the central nervous system. Adrenaline and noradrenaline are the fundamental neurotransmitters of the sympathetic nervous system (SNS).
There is evidence to suggest that CBG exerts influence on the CB2-receptor, but it not clear whether this effect is agonistic or antagonistic in nature.
Chemical properties of the CBG molecule
As with all cannabinoids, CBG is a hydrophobic lipid comprised of a 21-carbon chain attached to an aromatic hydrocarbon ring. The chemical formula of CBG is C21H32O2; that of THC is C21H30O2, as is that of CBC (cannabichromene) and CBD. CBC and CBD are both isomers of THC, meaning that they share the same molecular formula but have different structures.
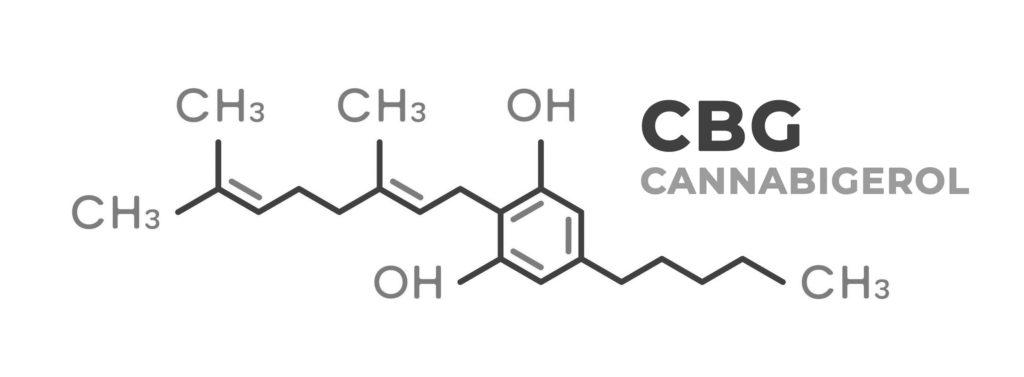
CBG is closely related to several other cannabinoids including cannabigerolic acid (C22H31O4), also known as CBGA, which is an important precursor for THC. CBG itself degrades to CBD and CBC, and it is also possible for CBG to degrade directly to THC, through the actions of the acid synthase enzyme unique to each cannabinoid (i.e. THC- CBD- and CBC-acid synthase). This process is relatively new to the scientific community, as it was previously believed that THC was an end-product of CBD degradation.
The CBG group of cannabinoids
The cannabigerol-type cannabinoid group contains seven related molecules including CBG itself. As well as CBG and CBGA, the CBG group contains cannabigerovarin (CBGV), cannabinerolic acid A (CBNA), cannabigerovarinic acid A (CBNVA), and the monomethyl esters of CBG and CBGA, CBGM and CBGAM respectively.

This group of cannabinoids is relatively understudied; however, it is believed that CBNA is also a precursor to THC in certain conditions. The molecular pathways through which THC is biosynthesised are still being discovered, and successful laboratory synthesis often occurs in different ways than those occurring in vivo. This partly explains why it was believed that THC is naturally synthesised from CBD, as until recently it was the only tested method for laboratory synthesis.
The CBG-dominant chemotype
In nature, the tendency for populations to tend towards a CBG-dominant chemotype (chemical phenotype) has been noted in certain regions, mostly in the high latitudes where hemp strains traditionally dominate. In ‘normal’ cannabis plants, CBG makes up no more than 10% of the cannabinoid profile while in CBG-dominant plants, the figure may be as high as 94%. In such populations, there is often a corresponding, remarkably low expression of THC, as little as 0.001% in some specimens.
It is believed that a recessive allele type at a single gene locus is responsible for this phenomenon. The locus in question, known simply as B, is thought to control the synthesis of THC or CBD from CBG. The CBG-dominant type has the allele B0, which encodes for a defective acid synthase, inactivating the process of synthesis and leading to unusually high CBG levels with correspondingly low THC.
The discovery of this phenomenon has led to improved understanding of the genetic mechanisms through which varieties that are dominant in just one cannabinoid can arise. Such varieties are potentially of great medicinal importance as they allow for easier extraction and study of the dominant cannabinoid.

Medicinal properties of CBG
Although CBG has not been extensively studied for its medical properties, several studies have been conducted. A 2009 study on glaucoma in cats concluded that both THC and CBG reduce intraocular pressure and increase aqueous outflow through the intricate mesh of drainage channels in the tissue surrounding the eye.
CBG has also been found to have an anti-nausea and anti-emetic (anti-vomiting) effect on rats, although this research has not been replicated in humans. Further, CBG has demonstrated anti-inflammatory effects on inflammatory bowel disease and colitis in mice. As well as this, CBG has also been shown to reduce pain, ease symptoms of psoriasis, act as an antibacterial agent and even slow the proliferation of tumour cells.
For these reasons, the medical significance of CBG continues to increase, with a growing number of cannabis cultivators focusing on growing strains of hemp that contain high levels of CBG. As this understanding grows, the mechanisms by which cannabis exerts its medical qualities on the human body becomes more complex and more profound.
- Disclaimer:This article is not a substitute for professional medical advice, diagnosis, or treatment. Always consult with your doctor or other licensed medical professional. Do not delay seeking medical advice or disregard medical advice due to something you have read on this website.










This is a very well written and informative article regarding CBG! Thank You! We have compiled a lot of information on CBG on our website should anyone want more information. Blessings and Thanks!
Non-Psychotropic Cannabinoid Halts Colon Cancer Progression
Thursday, 09 October 2014
Study: Administration Of Non-Psychotropic Cannabinoid Halts Colon Cancer Progression
Naples, Italy: The administration of the non-psychotropic cannabinoid cannabigerol (CBG) possesses potent anti-colon cancer activities and inhibits cancerous tumor growth, according to preclinical data published online ahead of print in the journal Carcinogenesis. Colorectal cancer is the third most common cancer in men and the second most common cancer in women worldwide.
Italian researchers at the University of Naples assessed the effects of CBG on colon tumorigenesis in vitro and in vivo. Investigators reported that the compound promoted apoptosis (cancer cell death) and reduced cell growth in colorectal cancer cells. CBG dosing also inhibited an experimental model of colorectal tumor growth in animals.
Authors concluded: “Our data show that the non-psychotropic cannabis ingredient cannabigerol inhibits the growth of colorectal cancer cells … and hinders the development and growth of colon carcinogenesis in vivo. … [W]e hypothesize that CBG may be a promising anti-colorectal cancer therapeutic agent, both for prevention and as a curative medicine.”
Cannabinoids and endocannabinoids have been shown both independently and adjunctively to possess anti-cancer activity in preclinical models, halting the proliferation of glioma cells, breast carcinoma, lung carcinoma, and lymphoma, among other cancer cell lines.
Full text of the study, “Colon carcinogenesis is inhibited by the TRPM8 antagonist cannabigerol, a cannabis-derived non-psychotropic ca
This is very interesting as it may block the interaction of alcohol & opiates from engaging with the receptor but more importantly it sounds like it may be a very effective bi-polar medication. From personal experience I get quick relief from sinus & nausea with the cannabis variety but to get a non euphoric medication. I get varying effects as a anti inflammatory,sometimes it is an anti inflammatory & sometimes it is an inflammatory, Overstimulating the nervous system.I a assume that it is because I don`t have scientifically controlled samples to examine them effectively. I don`t test hemp strains either. I also wonder if this new information is going to be lead to the development of effective non euphoric anti-consultants. Although it only requires very small regular doses to be effective & the subject becomes immune to the euphoric action of the drug. It also lasts a long time comparable to sustained release timing of standard pharmaceutical products in the case of seizure treatments.
Nice read.