Cannabinoids occur naturally in plants of the cannabis sativa L. species. We absorb them when we consume cannabis – depending on what exactly is consumed and how. The human body also produces them. In the future, we are likely to uncover further characteristics, benefits and forms of cannabinoids.
Cannabinoids are chemical compounds that affect cannabinoid receptors that exist in the human body, as well as in the organisms of certain other mammals. Cannabinoid receptors have a direct effect on the release of neurotransmitters in the brain. In other words, they influence the way in which information and chemical compounds are processed by the brain.
The main cannabinoid receptors that we know about have been christened CB1 and CB2. They are found in various areas of the brain and the nervous system, which means that they control a number of functions and can influence various aspects of our health (although in certain cases both receptors work together).
The currently best known and best loved cannabinoids are THC, known for its psychoactive properties, and also CBD, due to its potential medicinal benefits, which are increasingly being discovered. There are a wealth of other cannabinoids – so far research has identified 113 different cannabinoids.
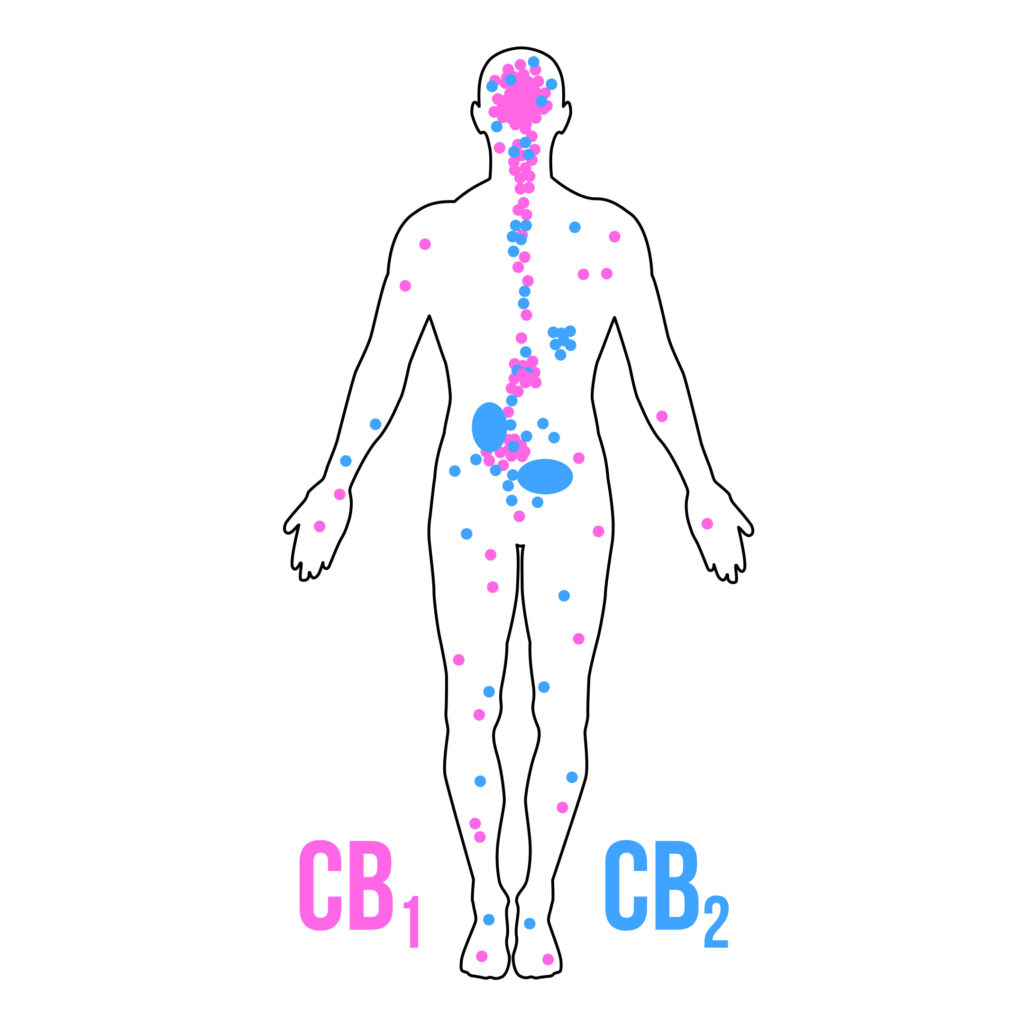
Where are cannabinoids found?
1. In plants
Cannabinoids exist in larger and smaller quantities in all the varieties of the Cannabis Sativa L. plant. Other plants also produce cannabinoids, but not in the same volumes (and not in the same way) as cannabis or hemp.
2. In the human body
Cannabinoids that are produced naturally by the human body are called endocannabinoids. The human network of cannabinoids and their corresponding cannabinoid receptors is referred to as the endocannabinoid system. It is essential for the human body to function correctly. A clinical lack of endocannabinoids is related to a whole series of illnesses and complaints. For example, cystic fibrosis may be caused by a malfunctioning endocannabinoid system.
What do cannabinoids do?
Early human development
Research has now shown that cannabinoids form a significant part of the primary functions of our body, right from the earliest stages of life. In fact, a well-functioning endocannabinoid system is necessary, for example, for the nidation of the fertilised ovum in the uterine lining, or to trigger a baby’s sucking reflex, for breast feeding. These are just a few of the many life-critical influences on the early development of human beings that are directly linked to the endocannabinoid system.

Neuroprotective properties and effects on appetite
Current research into illnesses such as dementia or other neurodegenerative diseases have suggested some neuroprotective effect of cannabis in animal models. A series of – mainly psychoactive – cannabinoids have been linked to neuroprotection, especially THC, THCA and possibly THCV. The last two have a very similar structure to THC.
THCV is well-known for being an appetite suppressant, a property also shared by other cannabinoids such as anandamide. Anandamide, an endogenous THC look-alike, is also well known, as is 2-AG, for interacting with CB receptors in the central and peripheral nervous system.

There is more…
There are many other properties that can be ascribed to THC, CBD and the other cannabinoids listed above: they can help with nausea, reduce inflammation, alleviate cramps etc. In fact, cannabinoids have already been studied in a variety of major impairments such as multiple sclerosis, Alzheimer’s, Parkinson’s, Tourette Syndrome, epilepsy, asthma and many others.
There is also some information about other cannabinoids that are not currently the focus of research. These include, for example, cannabinol (CBN), cannabigerol (CBG), cannabidivarin (CBV) and cannabicyclol (CBL). In most cases, these cannabinoids share features that have already been discovered in stronger cannabinoids (e.g. in the media favourites THC and CBD). For the moment, however, they are not being researched sufficiently, which means that it will be some time in the future before we find out more about these mysterious compounds!
Vaporizing
Vaporizing or steaming cannabis is, as far as we can tell, the best method of absorbing cannabinoids. In addition, this a healthier mode of consumption, which will suit most people, including those who are unable to smoke cannabis due to medical issues with their lungs.
Cannabinoid research versus prohibition
Cannabinoids form part of many different types of research. Sadly, due to the current illegality of the cannabis plant in most countries, it can be difficult to find plant material that can legally be used for research.

In a large number of countries, associations and even political parties are trying to encourage progress in cannabinoid research. Doctors also long for more information, more clinical tests and anything else that might help them to advise their patients better.
So far, however, just a handful of cannabinoid-based medicines are available to patients. While the legalisation of cannabis is moving forward at a rapid pace, at least compared to the previous decade, it could still take several more decades before things really change and cannabinoids are socially acceptable, without having some kind of stoner clichés weighing them down.
Until then, drug tests will continue to hit cannabis, which is why the question of how you can remove cannabis residues from your body is a very popular topic.
- Disclaimer:This article is not a substitute for professional medical advice, diagnosis, or treatment. Always consult with your doctor or other licensed medical professional. Do not delay seeking medical advice or disregard medical advice due to something you have read on this website.











Hi! I love how informative and great your articles are. Thanks a lot!
very interesting subject , great post.
Hi! Thought I,d drop a little note to explain how I am taking a dropper full of tincture cbd to battle severe fibromyalgia. I am sorry to say I am definitely allergic to it. I have allergies a mile long and trees and grasses are one of them. I am very disturbed by this because I firmly thought I found so real help. I am at a lose because I have pain everyWhere and I take
Medicine but does have any profound effect. Please Help!!!!!!! I need to know about synthetic cbd oil would this work for
Me and what company has it? Please answer,,,,,,,,help!!!!!!!!!!!
this
Dear Denise,
Thank you for your comment. We are sorry to hear about your situation. As Sensi Seeds is not a medical agency or practitioner, we cannot give any kind of medical advice other than to consult your registered healthcare professional. This article about the potential benefits of medicinal cannabis might be useful for you to show your healthcare provider if they are not familiar with it.
You may also find it helpful to contact a support group for medicinal cannabis patients. In the UK there is the United Patients Alliance, and throughout much of the rest of the world there is NORML, who should be able to put you in touch with a group in your area (search United Patients Alliance or NORML followed by your area name).
These are our pages on medicinal cannabis and medicinal cannabis strains, which you might also find interesting.
With best wishes,
Scarlet
Hi,
Thank you for your message. Please reach out to our Customer Service so they can check on your seeds. We do send replacement seeds in case of issues. It is likely there is something missing in your order information that prevented the package to get to you.
Kind Regards,
From experience of 48 years I know that Cannabis is a helpful and just fine.
I think this is a very good article, top notch! Thank you.
our dog has been on cbd oil for 4 weeks now what a difference it has made to his life ,before he could hardly walk, now he is down the beach or park every day walking and joining in the fun with other dog we just put 2 drops on his food twice a day cbd i would highly recomend thank you
Well I think it sounds good as I have MS and I used weed that could help me sleep and get back into eating. Cannabis is helping a lot of people that are sick although those that use it socially is still OK.
Where can i go To have cbd for fybromialgie 17/18.
From belgium have you adresses in holland? Coffeshops?
Hi Nathalie,
Please consult your doctor in order to know what is best for you.
Regarding CBD oil in general, Sensi Seeds offers the following: https://sensiseeds.com/en/cbd-products/cbd-oil
However, we are not health professionals, and therefore cannot advise you on which product is best for your condition.
With kind regards,
Hi there ,keep up the Good work …..am very interested in getting it right ????? which plant = which malady ….. Please advise …..lindsay …..hope all is well with yourselves .
The goverment /pharmacies companys do not want people to use cannibas as medicine.You can grow it in your own home an they cant make money of you. Greed is a sick reality
Here is a link to an article from 8 Nov 2016 about a 4 year clinical trial due to start on 1 Jan 2018 in DENMARK to evaluate cannabis for multiple sclerosis (spasticity, central pain and painful spasms: in addition permission to treat bladder function disorders can be considered); chronic pain (where other treatment is insufficient or not tolerated, e.g. neuropathic pain and “cancer” pain in joints, bones and muscles), spinal cord injuries (paraplegia: spasisticy, central pain and painful spasms); and chemotherapy-induced nausea and vomiting.(where other treatments have had no effect).
https://www.thelocal.dk/20161108/denmark-approves-medical-cannabis-trial
Could you please add the date to the comments so we know how old it is and to see if it is worth replying to?
Hi Nina,
The dates are already shown on comments in the bottom left hand corner, in dd/mm/yyyy format. The type is pale grey so possibly they are not showing up well if your screen brightness or contrast are turned down, or if you are viewing the site on a smaller screen? I hope this is helpful,
With best wishes,
Scarlet
I love this page, but I have read and re-read and am still confused. I’m seeing my Neurologist on the 16th of this month and I intend to ask her for medicinal Cannabis. If she goes along with the painstakingly job of starting the prosess of getting the permit and then writing the prescription I’m still not sure what to ask for. I have no hope she has been informed, so it’s all up to me. I have MS, Ulcerous Colitis and pain. I get Morphine-derivate patches for my pain, so it’s first and foremost the spasms I need help with. I’ve tried Sativex and for a short while I was able to run!!! Something that I haven’t done since I was at teacher’s College and that is more than 30 years ago. I was so happy and thought I’d found a good medicine, but a lass. After a week or so my stomach was so bad I had to give it up. Now I’m looking for something that helps me with my spasms without giving me a buzz or going through my intestinal tract. What should I ask for? It must be something I can smoke or use as a patch on my skin. Please advise me. Sincerely Torunn in Norway
Dear Torunn,
Thank you for your comment. We are sorry to hear about your situation. Unfortunately, as Sensi Seeds is not a medical practice, we are not able to provide any advice relating to medical situations other than to consult your doctor or other licensed medical professional, which you are already doing. There are also some support groups for medicinal cannabis users where you may be able to find more specific replies to your questions, or connect with people who are in a similar position. In the UK, there is the United Patients Alliance (you can find them on Facebook) and in the US and EU there are many branches of NORML (google NORML followed by your area name). We hope this is helpful.
With best wishes,
Scarlet
Hej Torunn!
You could have a look at Nordic Med Can AB´s product for vapourising on their Swedish website, or Endoca´s suppositories for rectal or vaginal use on the Danish website – 10 supps of 50 mg/supp for 49 euros. Organic raw cannabis sativa extract and coconut oil. There is a discount of 5% for 3 packets or 10% for 6 packets.
My wife Eileen’s life has been marred by Parkinson’s disease for two years. From being a confident intelligent outgoing person, once the financial director of a developing company, then running her own accountancy business, she has become withdrawn, depressed and increasingly disabled with nausea from reflux, pain in neck, arms and legs. Synamet bolstered by rasagiline medication serves to lessen symptoms but produce side effects that need further medication.
I have become a full time carer for a patient who is no longer able or willing to share new experiences and warm locations that would help her condition.
I am frustrated that there appears to be a medication that helps to alleviate the symptoms of Parkinson’s disease without producing nasty side effects that cannot be used in the UK because of a blanket ban on a drug marketed by criminals. This ban is blighting my life as well as my wife’s.
Thanks for the nice and well related information. I just would like to ask if you know of a country (out from Europe) where medical cannabis is free to obtain in order to cure myself of a thymus cancer and to avoid a third sternotomy. I live in Italy where laws for planting a cannabis seed of my own, is walking forward as a sick turtle…
More power.
A nice article.
My wife has m.s and suffers spasticity, cannot walk and can only stand for about 8 secs. She is 68 and said to me the other day ” i have missed out on middle age!! It annoys me very much that the british government seems to suffer from blinkered thinking ie canabis bad mutinational drugs good. We should be following the lead of the dutch government and investigating canabis as a medicine,urgently.My wife then may be able to entertain a ray of hope as at the moment she knows that she is only going to get worse.
…and Catalunya has no minister of Health,as it is not a country, but a region… even if they want independence.
Dear friend,
Even if Catalunya is not a country but a region, the government has a kind of health minister. It is called “Conseller” and he does exactly the same function. The “Departament de Salut” (the catalan health ministry) has created the Servei Català de la Salut (Catalan Health Service) and has consolidated a mixed health care system, with access to all health and social lines, giving response to the needs of health and people. This is something that has been like this for over 30 years and has nothing to do with the independence issue.
Thanks anyway for pointing out and we hope you keep reading Sensi Seeds articles,
This is an attack on our human rights that we are prohibited from using a medicine of our own choosing!
Your information on the Schengen Agreement and bringing prescribed cannabis into the UK is incorrect.
The test is not citizenship, it is residency. A UK citizen, resident elsewhere, who has a prescription in the country in which they are resident can bring prescribed cannabis into the UK.
Hello Peter,
Thank you for your comment and for pointing out this error. This article was originally written in German, and during the translation process there was a mistake caused by the double meaning of ‘citizen’ as passport holder / national and ‘citizen’ as resident of a town or city. The article has now been corrected to reflect the intended meaning.
Thanks for reading, and with best wishes,
Scarlet