Sativex by GW Pharmaceuticals is the first fully approved cannabis-based medicine. It is an oral spray containing THC and CBD, used in the treatment of multiple sclerosis (MS) patients. Today, Sativex is available in at least 25 countries, including Canada and many EU countries.
What is Sativex and who can use it?
Sativex is primarily marketed as an adjunctive treatment for spastic muscle paralysis associated with Multiple Sclerosis. On top of the ability to reduce severe spastic symptoms, Sativex also has been shown to effectively treat neuropathic symptoms and an overactive bladder in MS patients.
This product has been approved as MS medicine. Clinical trials up to Phase III were conducted in the US to prove whether it is also possible to use it as a supplement to treat tumour-related pain, especially for people who do not respond to high doses of opiates.
For now, the answer is no, as the CEO of GW, Justin Gover, explains: “Although we missed the primary endpoint in this trial, positive data from the Phase II programme make us confident that Sativex may effectively alleviate tumour-related pain in the patient population”.
Although marketed under the trade name Sativex, this medicinal product is commonly known as nabiximols. It is an extract made from two (absolutely secret) cannabis strains using ethanol and CO2.
Sativex is an oral spray (acting on the mucous membranes of the mouth) to be applied under the tongue. The product is essentially a cannabis oil tincture and contains THC and CBD in almost equal proportion: each spray contains a fixed dose of 2.7 mg THC and 2.5 mg CBD.
Sativex worldwide availability and distribution
In June 2010, Sativex was officially approved for the first time. Its launch in Canada occurred shortly after its market launch in the UK. This was the beginning of an unparalleled success story. Sativex is currently sold in at least 25 countries, including the Netherlands, Switzerland, Germany, France, Spain, Italy, Austria, Poland, Australia and New Zealand.
Sativex also appears to be close to approval in the US. After some delay caused by the COVID-19 pandemic, GW resumed its Phase III clinical trial. In a media release, Justin Gover was hopeful that Sativex will be approved as an official drug in the US market by the end of 2021.
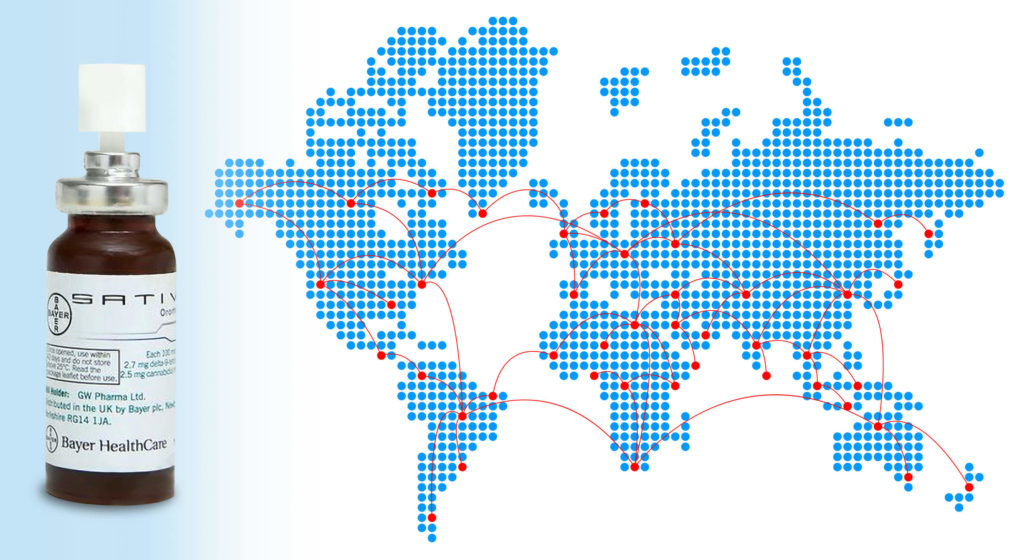
GW Pharmaceuticals is not the only player in the distribution of Sativex. In the UK, Sativex is distributed and marketed by the pharmaceutical giant Bayer, while Almirall owns distribution rights all over Europe. The annual report of this Spanish group shows sales of Sativex exceeding €30 million in 2019, around 20% more than in 2018.
What could apparently look like a gold mine to outsiders, actually falls short of expectations. In 2010, Piper Jaffray, a leading US investment bank, forecast sales of nearly $150 million (EU and Canada).
Natural cannabis flowers (or herbal cannabis, or buds), which are increasingly prescribed in Europe, are a thorn in the side of investors. In addition, Sativex is struggling with a price problem: in 2019, a monthly ration cost more than €500 in the UK. In Australia, the price of a six- to eight-week supply was the equivalent of more than €400.
History of Sativex – looking back
Cannabis is an ancient medicine and has been used as a remedy for thousands of years. However, it was only in the 1990s that scientists gradually began to understand how cannabis works in the body. The discovery of the endocannabinoid system was an important breakthrough and laid the foundation for further scientific progress.
GW Pharmaceuticals was the first company to seriously deal with the development of cannabis-based medicinal products. GW was founded in 1998 by Geoffrey Guy, a scientist who believed in the potential of cannabinoids.

Justin Gover came on board only one year later. As always happens, the early years were hard. It took more than 10 years before Sativex was launched on the market. However, each figure should be considered in relative terms: almost no medicine comes onto the market without a delay or setback.
Today GW produces 20 tonnes of cannabis per year, from which THC and CBD are extracted. Where exactly the cannabis is grown is top secret. The production facility is guarded around the clock and each plant is genetically fingerprinted to enable tracking, should anything be stolen.
Is Sativex less effective than herbal cannabis?
Sativex, manufactured by GW Pharmaceuticals, has been praised by some as a miracle drug, while others have dismissed it as mere quackery. The harshest critics have even gone so far as to claim that it is part of a far-reaching conspiracy to legitimise undercover medicinal cannabis. In this way, they say, pharmaceutical companies could profit from Sativex while ensuring that much cannabis research remains illegal.

But how about the effectiveness of Sativex? What do scientists say? Several studies have shown that Sativex effectively treats symptoms of spastic muscle paralysis associated with MS and neuropathies (nerve pain) caused by MS and cancer. Sativex enthusiasts argue that the added value of this medicine lies in the fact that, unlike herbal cannabis, it may be used for standardised treatments with fixed dosages.
However, Sativex obviously has no unique or special properties, apart from the fact that it can be professionally processed, packed, and sold in uniform doses. This is because Sativex is a simple cannabis tincture made with CO2 and ethanol and still contains all the terpenes and cannabinoids that can be found in any other cannabis extract. It may have specific medicinal effects associated with the strains used, but there is no steadfast evidence for this.
In addition, many criticise it for being slower and less effective than traditional methods of cannabis use. It is clear from patient surveys published online that people prefer to use herbal cannabis rather than Sativex. Normally, these patients find that Sativex is less effective in alleviating pain, although other surveys suggest that it may be very effective in reducing muscle spasms.
Arguments in favour of Sativex
We should not forget that Sativex and GW Pharmaceuticals have clearly proved to be powerful tools in the fight for the global recognition of medicinal cannabis. Sadly, the health systems of most Western countries today operate in such a way as to require a realistic approach to make a new drug available to the general public. This means using methods that grassroot activists usually do not like.
In these systems, cannabis has to be fragmented and processed into a medicinal product with a fixed dose for it to be considered compliant to the standards of modern medicine. GW Pharmaceuticals have now provided such a product, taken the necessary steps and carefully followed the rules of the game at every stage. In so doing, they have managed to make a cannabis-based medicine available in more than 25 countries worldwide, even in those lagging behind in medicinal cannabis legalisation.

From an ethical standpoint, it is absolutely reasonable to criticise the hypocrisy with which this company in the past pushed for legal reforms that could benefit it but were substantially of little use to home growers who were regarded as competitors damaging its business. However, companies like GW are also positively regarded for their strong lobbying activity in favour of reforms in countries that would otherwise resist them.
Whatever the final judgment on the future developments of GW Pharmaceuticals (with Sativex) will be, it is necessary to wait and see what the ultimate effect of the actions of this undertaking will be on the cannabis industry as a whole. The coming years will be decisive for the cannabis legalisation movement and, in this process, Sativex will surely play an important role.
Do you use Sativex in the treatment of MS? Or do you know someone who has been using Sativex? We are very interested to hear about your experience, especially if you are a herbal cannabis consumer. Tell us about it in the comments below, or write to us on Facebook. We are looking forward to hearing your comments!
- Disclaimer:This article is not a substitute for professional medical advice, diagnosis, or treatment. Always consult with your doctor or other licensed medical professional. Do not delay seeking medical advice or disregard medical advice due to something you have read on this website.












Wow! i never knew about this. Thank you!
Hey,
Thank you for your comment!
To learn a little more about medicinal cannabis, here are some links that you might find interesting.
What Is Medicinal Cannabis, and How to Choose a Medicinal Cannabis Strain? and Cannabis in the Netherlands – Laws, Uses and History
Thanks again, and I hope you continue to enjoy the blog.
With best wishes,
Mark